Detection of Vanadium Using Immobilizing 4-(2-pyridylazo)
| ✅ Paper Type: Free Essay | ✅ Subject: Chemistry |
| ✅ Wordcount: 2541 words | ✅ Published: 18 Jan 2018 |
“Naked eye detection of vanadium using immobilizing 4-(2-pyridylazo)-resorcinol on functionalized HMS”
Mohamad taghi Samadi1, Abas Afkhami2, Kadijeh Shamsi 1, *and Abdolmotaleb Said mohamadi1
abstract
In this work, inexpensive optical sensor for the naked eye detection of vanadium by immobilizing 4-(2-pyridylazo)-resorcinol on the functionalized hexagonal mesoporous silica via N-trimethoxysilylpropyl-N,N,N-trimethylammonium chloride in aqueous mediae has been used. The effects of several factors such as pH, reaction time, amount of the material, and the presence of various concentration HMS-TMAC-PAR has been studied so as to optimize operating conditions. The detection was result the color change of PAR complexation with vanadium as of orange-yellow to purple in alkaline solutions. Furthermore, the good performance of the hexagonal sensors was dependent on main factors such as, the reaction time, and the pH value that showed possible naked-eye detection of vanadium concentration of special interest was that our mesostructured sensor design supply control over the retention of the potential functionality of the naked-eye sensing system of vanadium ions upon the storage and even later several regeneration and reuse cycles, indicating wide-scale reversibility of sensing methods. The detection and quantification limits for the method when determining vanadium were 40 ppb–400 ppb, respectively, with a correlation coefficient of 0.99. Good chemical stability of the substantial was seeing for a period of several months.
Introduction
Vanadium is ordinarily distributed in the earth’s crust but in little profusion. Vanadium at trace amounts represents a vital element for normal cell growth, but can be toxic when present at much concentration(1). the combustion of fossil fuel are described as one of the important sources of environmental pollution by vanadium since coal and oil are in among the richest sources of the metal . Usual deposits of vanadium in coal and basic oil vary amid 1 and 1.5×103 mg/kg and it has been estimated that different 65×103tons are released into the environment every year by natural discharges such as volcanoes and 2×105 tons by anthropological resource(2). The concentration of vanadium in water is largely dependent on geographical position and ranges from 0.0002 to more than 0.1 mg/litre in freshwater(3). the selective and sensitive detection of low concentrations of vanadium is very important (4). Though several methods are available for detection (AAS/AES[1] ØŒICPMS[2] , ICPEAS[3] (1, 5) but these methods are expensive, need sophisticated apparatus’s and require extreme control of experimental procedures or unavailable for the on-site detection. The field of optical chemical sensors has been a growing research area over the last three decades. Chemical sensors are miniaturized devices that can supply real time and on-line information on the presence of specific compounds in even complex samples(6) In the last years,have seen the appearance of mesoporous silicate-based sensing uses. Sensor system Mesoporous silicas, with large BET surface area, narrowly distributed pore size, high porosity(7), have shown unlimited potential for sensing applications in new years. Morphological control gifts them versatility in the mode of arrangement whether as monoliths, mass powders, thin films, or implanted in coatings. Great surface areas and pore sizes greater has made them effective as adsorbent. This elude the leaching that can occur from physical encapsulation and mesopores can allow accessibility of analytes to many of active locates systems, UV-visible spectrophotometry has been often used for quantitative determination of analytes(8).
According to the recently research, The hexagonal mesoporous silica (HMS) is a material with high specific surface area and large pore diameter. HMS mesoporous silica has been first presented by Pinnavaia and co-workers(9) . present 0f high surface area and uniform pore size that make HMS suitable for catalyst supports(10) Moreover, Modifying the silica wall surface with organic groups to make their characteristic and achieve specific goal(11).
A inexpensive visual sensor for the optical detection of very low concentrations of many heavy metal based on functionalization with 4-(2-pyridylazo) resorcinol (PAR)(12). Its color changes from yellow to red while complexed with metal ions such as Cu2+. Various applications for PAR and its similarities have been put into practice. Therefore a different heavy metal ions responsive optical sensor (HMS-TMAC-PAR) has been prepared by grafted surface functionalization of the original HMS with (TMAC[4]) and then via immobilization of PAR probe. HMS-TMAC-PAR has some important advantages as a solid-state colorimetric sensor(13) .In the present paper, a simple, selective and sensitive method is proposed for the vanadium determination at very low concentrations.
2. Experimental
2.1.Reagents and materials
Tetraethylorthosilicate (TEOS) 98% (M = 208.33, d = 0.934 g/mL) were purchased from Sigma–Aldrich (Germany). dodecylamine (DDA) 98% (M = 185.36) was purchased from Merck, Germany. TMAC (50% methanol solution) was purchased from ABCR, Germany. PAR was purchased from Sigma–Aldrich (Germany) .Ethanol and toluene was purchased from Merck .All materials were of analytical reagent (AR) grade and used without further purification.
2.2. Apparatus and instruments
The XRD experiments were performed by a APD2000 diffractometer using Cu K_ radiation over the range of 1.54Aâ-¦.FTIR spectra of the samples were recorded in mid IR (400-4000 cm−1) region on a PERKIN EL MER65 spectrometer, using KBr pellet technique. UV–vis spectra were measured on a HACH DR -5000 spectrophotometer. In order to measure the pH value a HACH SENSION1 digital pH meter was used .
2.3.Synthesis of hexagonal mesoporous silica
Synthesis of hexagonal mesoporous silica was achieved by dissolve DDA (200 mg) in a mixed solvent of water (0.28 L) and ethanol (0.19 L), after that the addition of TEOS (0.0112 L). The pH value of the solution was adjusted to 9–11 with HCl. The finishing gel was stirred continually at room temperature for 18 h, and The product was filtered, washed by abundant distilled water, air-dried in an oven at 100â-¦C.and then was accomplished by calcinations at 640â-¦C for 4 h in air to remove the organic template.
2.4. Fabrication of HMS-TMAC-PAR
Anhydrous toluene containing TMAC and HMS was refluxed for 18 h under an N2 atmosphere. The reaction mixture was washed thoroughly with ethanol and water to remove unreacted TMAC and then material was dried in an oven at 65 â-¦C for 12 h to obtain HMS-TMAC. The immobilization of PAR on HMS-TMAC was performed by the addition of 0.6 g HMS-TMAC solid to PAR solution (93 × 10−6 M, 180 mL) in stirring for 3 h. it was washed with water to remove unreacted PAR. The material was dried in a vacuum oven at 65 °C overnight and then ground to fine powder. The synthetic routes of HMS-TMAC-PAR were illustrated in Scheme 1.

Scheme 1. HMS-TMAC-PAR
2.5.Detection procedure
Batch and column processes were employed to study the detection of vanadium from aqueous solutions with HMS-TMAC- PAR. Concentration of stock solutions of vanadium (V) (1000.00 µg ml-1.) Prepared by dissolving 2.2965g ammonium metavanadate (99.99% Merck) in 1000ml of 5% hydrochloric acid solution. The suspension solutions of HMS-TMACPAR (0.5 g L−1) were prepared in aqueous solution at different pH values, which were adjusted with 0.2 M NaOH or 0.2 M HCl. Every time a 2 mL suspension solution of HMS-TMAC-PAR was filled in a 1 cm quartz cell and various stock solutions of cations were added into the quartz cell gradually by using a micro-syringe. Standard and blank solutions were prepared, in all the experiments in the same way as those samples. The linear working range for measurements was (y = 0.005x + 0.015, R2 = 0.990).
The vanadium was estimated qualitatively using naked-eye colorimetric assessment and it was detected quantitatively using UV–vis reflectance spectrometry at 510 nm.
3.Results and discussion
3.1.Characteristics of HMS-TMAC-PAR
The XRD patterns of samples are displayed in Fig. 1. The XRD measurements were employed in order to study the mesoporous structure of the supports. the patterns have a single low-angle diffraction peak at about 2 θ =1.9â-¦ for HMS and HMS-TMAC- PAR.
but, the intensities of the peak of sample HMS-TMAC- PAR decrease, suggestive of that the mesoporous structure of sample HMS-TMAC- PAR. happen to fewer uniform ahead the foreword of the organic molecules interested in HMS(13).

Figure 1. XRD patterns of HMS and HMS-TMAC- PAR.
FT-IR spectra of HMS-TMAC-PAR is shown in Fig. 2. The HMS-TMAC-PAR sample show the characteristic Si- O- Si bands associated by the shape of a condensed silica network that be present at 1091, 965, 799 and 459 cm−1. The findings revealed that the mesoporous silica (HMS) has been grafted via TMAC organic molecules(13).

Fig. 1. FT-IR spectra of (a) HMS, and (b) HMS-TMAC-PAR.
3.2.Calibration graph
The effects of various factors such as pH, reaction time, amount of material needed, and the presence of various concentration of vanadium were studied to optimize the conditions for quick visual detection of vanadium.
Fig..3represents the absorbance of HMS-TMAC-PAR at 510 nm against vanadium concentrations and shows a linear correlation in the range from 40 to 500 ppb with a correlation coefficient of 0.99 . The nonlinear nature of the curve at higher concentrations is due to saturation effects.

Fig.3.Calibration curves of the absorbance of HMS-TMAC-PAR with different concentrations of vanadium at pH 12.0 at 510 nm.
The detection limit of HMS-TMAC- PAR for vanadium was also tested. Fig. 3 represents the absorbance of HMS-TMAC-PAR at 510 nm against vanadium concentrations and a good linearity relationship was gained in the range of 40 to 500 ppb with a correlation coefficient of 0.99. The spectrometer detection limit (DL) of vanadium using HMS-TMAC-PAR was estimated from the linear part of the calibration plot, according to the Eq. (1)
DL =  (1)
(1)
where k is a factor equal to 3, m is the slope of the calibration graph in the lin-ear range, and Sb is the standard deviation for the blank. Here, The accuracy and precision of the method were tested by five successive measurements using a standard solution of 200ppb of vanadium. The resultant DL value is 0.88ppb.
3.3. Effect of the pH
In order to determine the pH effect on the detection capacity of HMS-TMAC- PAR in batch experiments, solutions were prepared at different pH levels ranging from 2, 7 and 10.
The pH-dependence studies showed that the complexation of vanadium with HMS-TMAC- PAR is formed in a wide range of pH, from 2 to 10(Fig. 4), which is similar to the results reported by Gang Li et al(13) . Also displays that upon the addition of vanadium, the virtual maximum absorption at pH 10.0, which has been selected as the specific pH for detecting vanadium in aqueous solution using HMS -TMAC- PAR. Hence, the maximum color and spectrum development of [V–HTP]n+ complexes were at a pH value of 10.
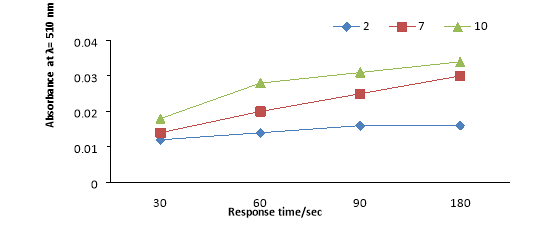
Fig. 4. Effect of pH on absorbance at 510 nm in UV–vis spectra of HMS-TMAC-PAR (0.5 g L−1 ) with vanadium 200ppb.
3.4. Effect of the reaction time
Studies on the reaction time indicated that the kinetic time response of the V–HTP complex formation was studied by constantly monitoring the absorption spectra .as shown in Fig. 5. After addition of 200ppb of vanadium, the absorbance increases rapidly within 90 s and in that case residue constant with time, so shows that the reaction has completed. . Therefore HMSTMAC- PAR had been can suplid rapid colorimetric detection.

Fig. 5. Response time of HMS-TMAC-PAR (0.5 g L−1, pH 12.0) with vanadium 200ppb
the later addition of vanadium ions, the color change of the sensors as a function of time. The findings revealed that charge transfer among the vanadium and PR probe has been accomplished in a short time (30 s ≤ Response time ≥180s) caused by the aptitude of the nanostructured sensors with open, great, and identical pore architectures to let the able binding of vanadium ions with graft PR-probe. actually the high metal change, that is ion transport, and the similarity of the metal–ligand binding were significantly affected by the structural morphology and geometry, as obviously evidence from the Response time value(14).
3.5. Effect of sensor amount
Studies on the sensor amount (Fig. 6) indicated that In actual fact, porosity, high surface area of the HMS hexagonal materials are beneficial to permit high adsorption capability of the chromophore investigation in quick kinetic systems. The amount adsorbed of the probe have a important roul in making a significant ion-sensing system. but , results show that the nature of the ion-sensing system is also reliant on the amount of solid support-probe used. On the other hand, the PR probe concentration significantly led to better sensitivity of the chemo sensors. The results show that 0.5 gr of sensors was enough to monitor a respectable color separation between the blank and the sample even at low vanadium ion concentration.

Fig. 6. Effect of HMS-TMAC-PAR sensor amounts with vanadium (200 ppb) at pH 10.0, 510 nm
3.6. Effect concentration of vanadium
In order to evaluate the effect of initial vanadium concentration, The naked-eye detection is achieved at a concentration range of 40–400 ppb using HMS-TMAC-PAR Fig.7. A considerable color change for HMS-TMAC-PAR is observed from yellow to purple with an increase in vanadium concentration
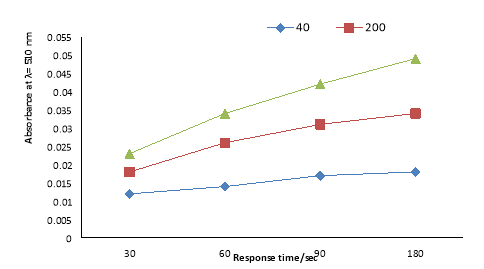
Fig. 7. Color reaction of HMS-TMAC-PAR (0.5 g L−1, pH 10.0) with various concentrations of vanadium
conclusions
A highly sensitive optical sensor for the visual detection of low concentrations of vanadium is designed by the immobilization of PAR on the functionalized HMS. The high sensitivity of the optical sensor could be caused by its high surface area and the uniform pore sizes and pore shapes of the mesoporous silica. Controlled experimental conditions also allowed naked-eye detection of vanadium ion concentration with detection limit as low as 0.88ppb In alkaline solutions .generally, HMS-TMAC-PAR provide strange sensitivity, reusability and fast colorimetric detection of heavy metal ions, particularly vanadium for track of heavy metal pollutants in the environmental samples. The findings revealed that it could be successfully used as a colorimetric optical sensor for the quantitative and qualitative detection of very low concentrations of vanadium in various samples.
REFERENCES
- PyrzyØ¥„ska K, Wierzbicki T. Determination of vanadium species in environmental samples. Talanta. 2004;64(4):823-9.
- Adachi A, Sawada S, Shida K, Nakamura E, Okano T. Determination of vanadium in foods by atomic absorption spectrophotometry. Science and Technology of Advanced Materials. 1999;5(1):15-21.
- Full TOC. 6.12 Vanadium. Changes.523:80.
- Hiraki K, Nakaguchi Y, Morita M, Kitamaki H. Determination of trace vanadium by addition of both sodium selenite and calcium chloride with polarized Zeeman effect graphite furnace atomic absorption spectrometry. Analytical sciences. 1986;2:561-6.
- Filik H, Berker KI, Balkis N, Apak R. Simultaneous preconcentration of vanadium (V/IV) species with palmitoyl quinolin-8-ol bonded to amberlite XAD 2 and their separate spectrophotometric determination with 4-(2-pyridylazo)-resorcinol using CDTA as masking agent. Analytica chimica acta. 2004;518(1):173-9.
- McDonagh C, Burke CS, MacCraith BD. Optical chemical sensors. Chemical reviews. 2008;108(2):400.
- Morante-Zarcero S, Snchez A, Fajardo M, del Hierro I, Sierra I. Voltammetric analysis of Pb (II) in natural waters using a carbon paste electrode modified with 5-mercapto-1-methyltetrazol grafted on hexagonal mesoporous silica. Microchimica Acta.169(1-2):57-64.
- Melde BJ, Johnson BJ, Charles PT. Mesoporous silicate materials in sensing. Sensors. 2008;8(8):5202-28.
- P.T. Tanev, M. Chibwe, T.J. Pinnavaia, Titanium-containing mesoporous molecular sieves for catalytic oxidation of aromatic compounds, Nature 368 (1994)321–323.
- Martn N, Lpez-Gaona A, Viniegra M, Villamil P, CØ£rdoba G. Synthesis and characterization of a mesoporous HMS and its use as support of platinum catalysts. Reaction Kinetics, Mechanisms and Catalysis.101(2):491-500.
- Aguado J, Arsuaga JM, Arencibia A, Lindo M, Gascn V. Aqueous heavy metals removal by adsorption on amine-functionalized mesoporous silica. Journal of Hazardous Materials. 2009;163(1):213-21.
- Balaji T, Sasidharan M, Matsunaga H. Naked eye detection of cadmium using inorganic-organic hybrid mesoporous material. Analytical and bioanalytical chemistry. 2006;384(2):488-94.
- Liu T, Li G, Zhang N, Chen Y. An inorganic-organic hybrid optical sensor for heavy metal ion detection based on immobilizing 4-(2-pyridylazo)-resorcinol on functionalized HMS. Journal of Hazardous Materials.201-“202(0):155-61.
- Zhang N, Li G, Cheng Z, Zuo X. Rhodamine B immobilized on hollow Au-HMS material for naked-eye detection of Hg 2+ in aqueous media. Journal of hazardous materials. 2012;229– 230(2012):404-10.
[1] -Atomic emission spectroscopy
[2]– Iinductively coupled plasma mass spectrometry
[3]-Inductively Coupled Plasma Atomic Emission Spectroscopy
[4] – N-trimethoxysilylpropyl-N,N,N-trimethylammonium chloride
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal



